|
hc8meifmdc|20005939267D|healthm_live|health_library|health_library_details|0xfdff79be010000006b02000001000100
| Over View |
Glucosamine is composed of glucose (a sugar) and glutamine (an amino acid). It is utilized by chondrocytes to form glycosaminoglycans (GSG) and proteoglycans (PG). Both of these constituents attract and bind water into cartilage, increasing resiliency. Research indicates that glucosamine assists the body in repairing damaged, eroded cartilage tissues. Many studies have been conducted on glucosamine sulfate and glucosamine hydrochloride, with a preponderance of positive results. Glucosamine sulfate is considered as the more effective of the two.
Chondroitin is classified as a glycosaminoglycan (GAG). It bonds with collagen to form the basis of connective tissue. Chondroitin helps attract fluid into proteoglycans, thereby bringing nutrients into cartilage and providing shock absorption. While glucosamine helps manufacture and maintain cartilage, chondroitin keeps cartilage from becoming malnourished. Chondroitin works synergistically with glucosamine, and together, they form the basis of many of the industry’s most popular joint support supplements.
Glucosamine, chondroitin sulfate, and MSM (methylsulfonylmethane) occur naturally in the body. The glucosamine used in supplements is typically derived from the shells of shrimp or crabs although a corn source is also available. Glucosamine is available in a variety of chemical forms, such as glucosamine hydrochloride and glucosamine sulfate. These vary in terms of the amount of actual glucosamine available for use in the body from an equal weight of each form. Chondroitin sulfate is generally derived from pig or cow cartilage, but even shark and chicken cartilage has been used. Chondroitin-like mucopolysaccharides from algae are also available. MSM is synthetically produced.
What is Glucosamine and Chondroitin?
|
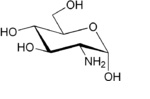 |
| (3R,4R,5S)-3-Amino-6-(hydroxymethyl)oxane-2,4,5-triol |
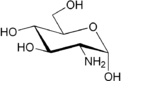 |
| (3R,4R,5S)-3-Amino-6-(hydroxymethyl)oxane-2,4,5-triol |
|
|
The key element in human joints is articular cartilage, the shockabsorbing tissue that connects two bones together. Articular cartilage is comprised of two different molecules, collagen and proteoglycans, with the remainder composed primarily of water (65-85%). Collagen, a protein that binds tissue together, provides elasticity. Proteoglycans, composed of sugars and protein, absorb water, which provides lubrication and resiliency, nature’s shock absorber for your joints. Both compounds are produced by chondrocytes, caretaker cells responsible for the formation and maintenance of cartilage. A deficiency in any one of the above constituents will increase the likelihood of wear and tear on articular cartilage, which can eventually lead to compromised joint function. |
|
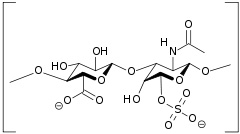 |
| Chondroitin sulfate - alternate diagram |
|
Glucosamine and chondroitin sulfate supplements are used to slow the progression of osteoarthritis — the deterioration of cartilage between joint bones — and to reduce the associated pain. A study sponsored by the National Institutes of Health (NIH) called the Glucosamine/Chondroitin Arthritis Intervention Trial (or GAIT study), showed that the combination of glucosamine and chondroitin sulfate seems to be effective in osteoarthritis patients with moderate to severe knee pain, although not those with mild pain. However, a 2007 review of several past, large-scale studies of chondroitin alone concluded that it did not provide a benefit.
A well-controlled study of glucosamine sulfate (1,500 mg per day) versus placebo for lower back pain caused by osteoarthritis showed that both glucosamine and placebo improved symptoms reported by patients. However, there was no significant difference in the level of improvement between the groups, suggesting that glucosamine alone is not truly effective for lower back pain (Wilkens, JAMA 2010).
Glucosamine is thought to promote the formation and repair of cartilage. Chondroitin is believed to promote water retention and elasticity in cartilage and inhibit enzymes that break down cartilage. In veterinary medicine, glucosamine and chondroitin, often in combination, are commonly used in the treatment of degenerative joint disease — typically in older dogs and cats as well as in horses.
How is it Made?
Glucosamine is a compound found naturally in the body, made from glucose and the amino acid glutamine. Glucosamine is needed to produce glycosaminoglycan, a molecule used in the formation and repair of cartilage and other body tissues. Production of glucosamine slows with age.
Where is it Found?
Glucosamine is most commonly obtained from the shell portion of shellfish, and the chondroitin is from the cartilage of animals such as pig’s ears or nose, and most often from shark cartilage.
|
|
|
|
|
Benefits / Uses
Glucosamine chondroitin MSM improves the consistency of the synovial or lubricating fluid in joints. It reduces the swelling in the joint and decreases the associated pain. It enhances the body's ability to repair cartilage and other connective tissues and improves flexibility. The effects of the three supplements working together is greater than if only one or two compounds were taken. When used with conventional anti-inflammatory medications such as ibuprofen (Advil, Motrin, etc.), chondroitin sulfate seems to help reduce hip and knee joint pain due to a type of arthritis known as osteoarthritis. It can take up to 4 months of treatment for relief of pain.
Effective for:
Dry eyes as an eye drop.
Reducing pain from a kind of arthritis called osteoarthritis.
In osteoarthritis, the cartilage in the joints breaks down. Taking chondroitin sulfate, one of the building blocks of cartilage, might slow this breakdown.
A well-controlled study of glucosamine sulfate (1,500 mg per day) versus placebo for lower back pain caused by osteoarthritis showed that both glucosamine and placebo improved symptoms reported by patients. However, there was no significant difference in the level of improvement between the groups, suggesting that glucosamine is not truly effective for lower back pain
Dosage
BY MOUTH:
For osteoarthritis: the typical dose of chondroitin sulfate is 200-400 mg two to three times daily or 1000-1200 mg as a single daily dose.
APPLIED TO THE SKIN:
For osteoarthritis: a cream containing 50 mg/g of chondroitin sulfate, 30 mg/g of glucosamine sulfate, 140 mg/g of shark cartilage, and 32 mg/g of camphor has been used as needed for sore joints for up to 8 weeks.
Possible Side-Effects / Precautions / Possible Interactions
Chondroitin sulfate seems to be safe for most people. It can cause some mild stomach pain and nausea. Other side effects that have been reported are diarrhea, constipation, swollen eyelids, leg swelling, hair loss, and irregular heartbeat.
There is some concern about the safety of chondroitin sulfate because it comes from animal sources. Some people are worried that unsafe manufacturing practices might lead to contamination of chondroitin products with diseased animal tissues including those that might transmit bovine spongiform encephalopathy (mad cow disease). So far, there are no reports of chondroitin causing disease in humans, and the risk is thought to be low.
Some chondroitin products contain excess amounts of manganese. Ask your healthcare professional about reliable brands. Do not use chondroitin sulfate if:
|
|
 |
You are pregnant or breast-feeding. |
 |
You have asthma. |
 |
You have prostate cancer or an increased risk for prostate cancer. | |
|
|
Interactions
Warfarin (Coumadin) is used to slow blood clotting. There are several reports showing that taking chondroitin with glucosamine increases the effect of warfarin (Coumadin) on blood clotting. This can cause bruising and bleeding that can be serious. Don't take chondroitin if you are taking warfarin (Coumadin).
Research Studies / References
|
 |
Baeurle SA, Kiselev MG, Makarova ES, Nogovitsin EA (2009). "Effect of the counterion behavior on the frictional–compressive properties of chondroitin sulfate solutions". Polymer 50: 1805–1813. doi:10.1016/j.polymer.2009.01.066. |
|
|
 |
Wandel S, Jüni P, Tendal B, Nüesch E, Villiger PM, Welton NJ, Reichenbach S, Trelle S (2010). "Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: network meta-analysis". British Medical Journal 341: c4675. doi:10.1136/bmj.c4675. PMID 20847017. Free PDF online |
|
|
 |
P. A. Levene and F. B. La Forge (1913). "On Chondroitin Sulphuric Acid". J. Biol. Chem. 15: 69–79. Free PDF online |
|
|
 |
MeSH Chondroitin+sulfates |
|
|
 |
Davidson EA, Meyer K (1954). "Chondroitin, a new mucopolysaccharide". J Biol Chem 211 (2): 605–11. PMID 13221568. Free PDF online |
|
|
 |
Barnhill JG, Fye CL, Williams DW, Reda DJ, Harris CL, Clegg DO (2006). "Chondroitin product selection for the glucosamine/chondroitin arthritis intervention trial". J Am Pharm Assoc (Wash DC) 46 (1): 14–24. PMID 16529337. |
|
|
 |
Silbert JE, Sugumaran G (2002). "Biosynthesis of chondroitin/dermatan sulfate". IUBMB Life 54 (4): 177–86. doi:10.1080/15216540214923. PMID 12512856. |
|
|
 |
Vergés J, Castañeda-Hernández, G. On the bioavailability of oral chondroitin sulfate formulations: proposed criteria for bioequivalence studies. Proc. West. Pharmacol. Soc., 2004; 47: 50-53 |
|
|
 |
Adebowale AO Cox DS, Liang Z, Eddington ND (2000). "Analysis of glucosamine and chondroitin sulfate content in marketed products and the Caco-2 permeability of chondroitin sulfate raw materials". J Am Nutr Assoc 3: 37–44. http://www.americanutra.com/itemdetail.cfm?ProductID=37.[dead link] |
|
|
 |
Jordan KM, Arden NK. EULAR Recommendations 2003: an evidence based approach to the management of knee osteoarthritis: Report of a Task Force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Ann Rheum Dis, 2003; 62:1145–1155 |
|
|
 |
Forsyth R, Brigden C, Northrop A (2006). "Double blind investigation of the effects of oral supplementation of combined glucosamine hydrochloride (GHCL) and chondroitin sulfate (CS) on stride characteristics of veteran horses". Equine veterinary journal. Supplement (36): 622–5. PMID 17402494. |
|
|
 |
Jamie G. Barnhill, Carol L. Fye, David W. Williams, Domenic J. Reda, Crystal L. Harris, and Daniel O. Clegg. Chondroitin Product Selection for the Glucosamine/Chondroitin Arthritis Intervention Trial. J Am Pharm Assoc. 2006; 46:14–24. |
|
|
 |
Ji D, Roman M, Zhou J, Hildreth J (2007). "Determination of Chondroitin Sulfate Content in Raw Materials and Dietary Supplements by High-Performance Liquid Chromatography with Ultraviolet Detection After Enzymatic Hydrolysis: Single-Laboratory Validation". J AOAC Int. 90 (3): 659–669. PMID 17580617. |
|
|
 |
Hathcock JN, Shao a. Risk assessment for glucosamine and chondroitin sulfate. Regulatory Toxicology and Pharmacology, 2007; 47: 78-83 |
|
|
 |
Andermann G, Dietz M. The influence of the route of administration on the bioavailability of an endogenous macromolecule: chondroitin sulfate (CSA). Eur J Drug Metab Pharmacokinet 1982;7:11–6 |
|
|
 |
Conte A, Palmieri L, Segnini D, Ronca G. Metabolic fate of partially depolymerized chondroitin sulfate administered to the rat. Drugs Exp Clin Res. 1991;17:27-33. |
|
|
 |
Conte A, de Bernardi M, Palmieri L, Lualdi P, Mautone G, Ronca G. Metabolic fate of exogenous chondroitin sulfate in man. Arzneimittelforschung. 1991;41:768-72. |
|
|
 |
Conte A, Volpi N, Palmieri L, Bahous I, Ronca G. Biochemical and pharmacokinetic aspects of oral treatment with chondroitin sulfate. Arzneimittelforschung. 1995;45:918-25. |
|
|
 |
Palmieri L, Conte A, Giovannini L, Lualdi P, Ronca G. Metabolic fate of exogenous chondroitin sulfate in the experimental animal. Arzneimittelforschung. 1990;40:319-23. |
|
|
 |
Ronca F, Palmieri L, Panicucci P, Ronca G. Anti-inflammatory activity of chondroitin sulfate. Osteoarthritis Cartilage. 1998;6 Suppl A:14-21. |
|
|
 |
Monfort J, Pelletier J-P, Garcia-Giralt N, Martel-Pelletier J. Biochemical basis of the effect of chondroitin sulfate on osteoarthritis articular tissues. Ann Rheum Dis 2007; doi:10.1136/ard.2006.068882 . |
|
|
 |
Jomphe C, Gabriac M, Hale TM, Heroux L, Trudeau LE, Deblois D, Montell E, Verges J, du Souich P. Chondroitin Sulfate Inhibits the Nuclear Translocation of Nuclear Factor-kappaB in Interleukin-1beta-Stimulated Chondrocytes. Basic Clin Pharmacol Toxicol. 2007 Nov 5 |
|
|
 |
Kwan Tat S, Pelletier JP, Verges J, Lajeunesse D, Montell E, Fahmi H, Lavigne M, Martel-Pelletier J. Chondroitin and glucosamine sulfate in combination decrease the pro-resorptive properties of human osteoarthritis subchondral bone osteoblasts: a basic science study. Arthritis Res Ther. 2007 Nov 9;9(6):R117 |
|
|
 |
Bourgeois F, Chales C, Delais J, Delcambre B, Kuntz JL, Rozenber S. Efficacy and tolerability of chondroitin sulfate 1200 mg/day vs chondroitin sulfate 3x400 mg / day vs placebo. Osteoarth Cart 1998;6 Suppl A:25-30. |
|
|
 |
Bucsi L, Poor G. Efficacy and tolerability of oral chondroitin sulfate as a symptomatic slow acting drug for osteoarthritis (SYSADOA) in the treatment of knee osteoarthritis. Osteoarth Cart 1998;6 Suppl A:39-46. |
|
|
 |
Uebelhart D, Thonar EJMA, Delmas PD, Chantraine A, Vignon E. Effects of oral chondroitin sulfate on the progression of knee osteoarthritis: a pilot study. Osteoarth Cart 1998;6 Supp A: 39-46. |
|
|
 |
Pavelka K, Bucsi L, Manopulo R. Double-blind, dose effect study of oral CS 4&6 1200 mg, 800 mg, 200 mg against placebo in the treatment of femoro-tibial osteoarthritis. Litera Rheumatol. 1998, 24: 21-30. |
|
|
 |
Morreale P, Manopulo R, Galati M, Boccanera L, Saponati G, Bocchi L. Comparison of the anti-inflammatory efficacy of chondroitin sulfate and diclofenac sodium in patients with knee osteoarthritis. J Rheumatol 1996;23:1385-91. |
|
|
 |
Uebelhart D, et al. Intermittent treatment of knee osteoarthritis with oral chondroitin sulfate: a one-year, randomized, double-blind, multicenter study versus placebo. Osteoarthritis Cartilage. 2004 Apr;12(4):269-76. |
|
|
 |
Leeb BF, et al. A metaanalysis of chondroitin sulfate in the treatment of osteoarthritis. J Rheumatol. 2000 Jan;27(1):205-11. | |
| |
|